AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
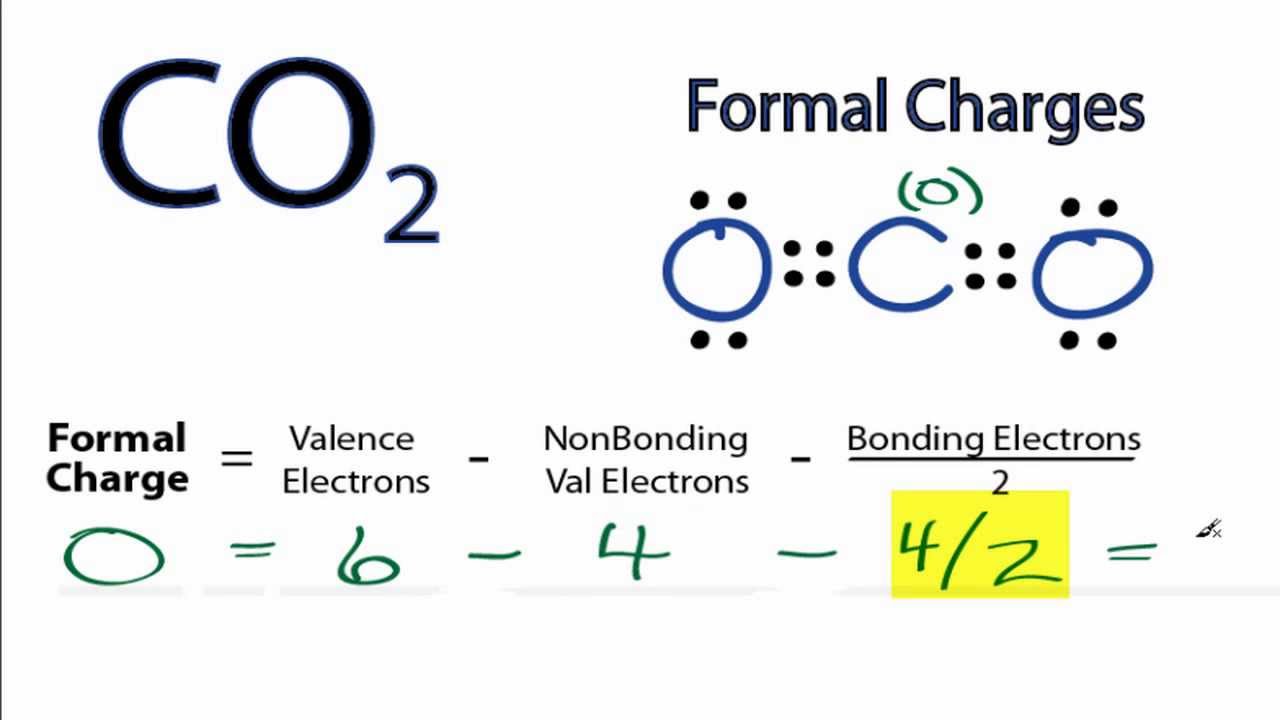
Calculating formal charge.9/23/2023 Let’s make sure you understand this shortcut This shortcut is guaranteed to save precious seconds on your exam IF AND ONLY IF you understand how to apply it.īut when you understand it you’ll be able to solve formal charge in your head, in under 8 seconds per atom. Here another option, MY version – one that is easier, faster, and comes out with the same result! Formal Charge Calculation Shortcut Yes the equation works, but it’s far too tedious and annoying! There are just too many steps and calculations. A triple bond of 6 electrons allows each atom to claim three.ĭo you dread running through this every time you finish a reaction? A double bond containing four electrons allows each atom to claim 2. ½ # bonding electrons = Since a bond is formed by sharing 2 electrons between 2 atoms, every atom in the bond can only take credit for one of the 2 electrons.Each electron counts as one and so a pair counts as two. Lone Pairs = lone electrons sitting on the atom. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed